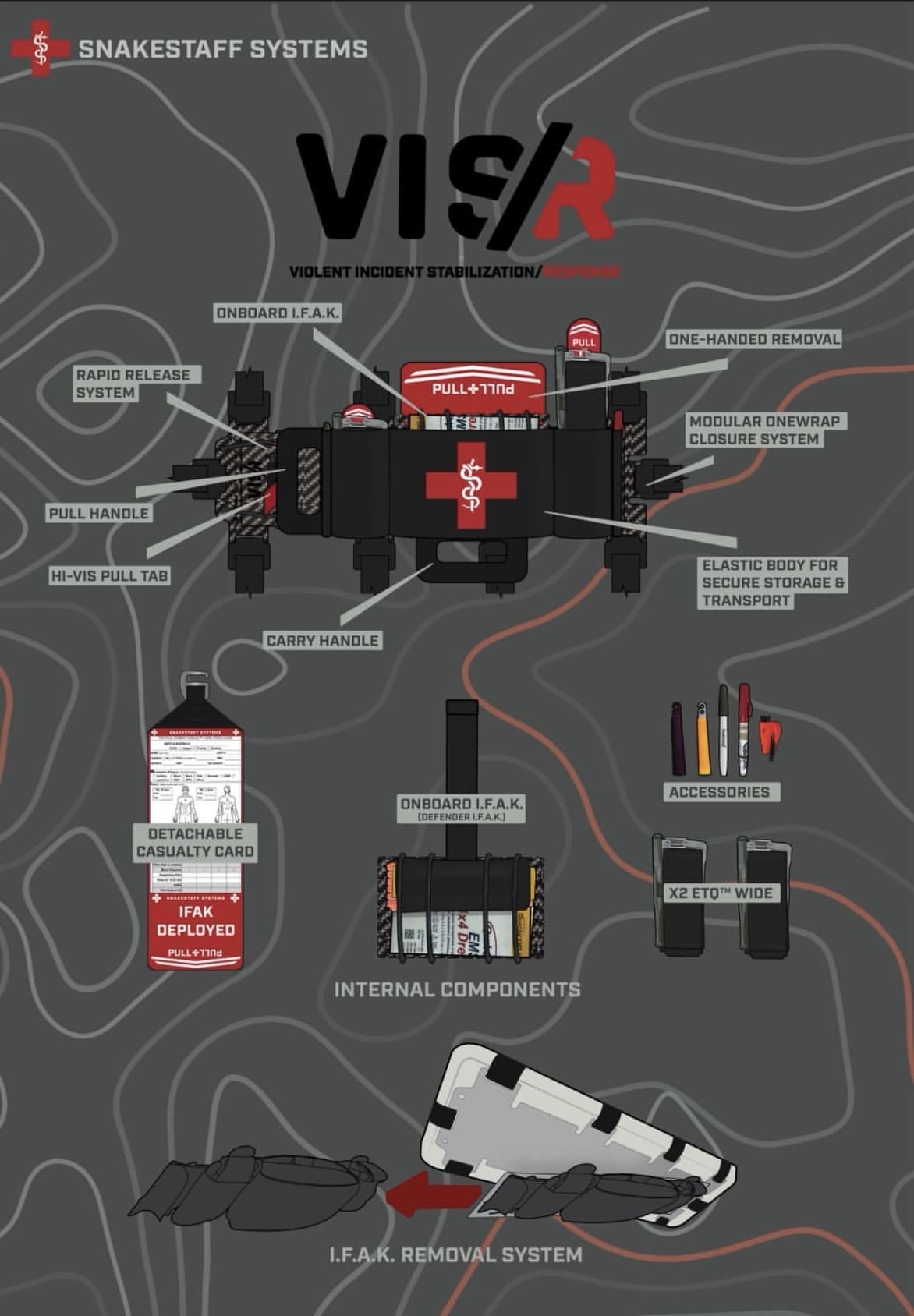
SAN DIEGO — The first face the Artemis II crew will see upon their return to Earth will be the face of a U.S. Navy Sailor.

Lt. Cmdr. Jesse Wang, Senior Chief Hospital Corpsman Laddy Aldridge, Chief Hospital Corpsman Vlad Link, and Hospital Corpsman 1st Class Steve Kapala make up the dive medical team who will be the first to open the Orion capsule upon its return to Earth, make initial medical assessments of the Artemis II crew, and assist them out of the capsule safely and efficiently.
They will provide initial one-on-one assistance to Christina Koch, Jeremy Hansen, Reid Wiseman, and Victor Glover respectively. Training for this mission has been a years-long process between the four team members as the first-contact medical providers inside the capsule.
Often working in expeditionary warfare communities, Navy dive medical personnel are certified divers and undergo specialized training, making them experts in decompression illnesses and other undersea medical considerations. Their mission is to care for and ensure dive-qualified service members are safe to conduct diving operations.
Following Orion’s reentry into Earth’s atmosphere and splashdown in the Pacific Ocean, the medical team will enter the capsule to conduct initial exams for the crew, provide triage care as necessary, and assist the astronauts in egress onto the inflatable raft set up outside by Navy divers. The first-contact medical providers will then prepare the crew to be airlifted by Helicopter Sea Combat Squadron (HSC) 23 back to amphibious transport dock USS John P. Murtha (LPD 26) for follow on evaluations.
Wang, assigned to Explosive Ordnance Disposal Group 1, serves as the lead for the four-man team. From Laguna Beach, Calif., he is a board-certified emergency medicine doctor by trade, having completed residency training at Lincoln Hospital in Bronx, New York. He joined the Navy in 2021 and was designated as an undersea medical officer in 2024.
“As a proud member of the undersea medical community, I am particularly humbled to play a part in this mission,” Wang said. “It is the honor of a lifetime to stand here today, ready to provide the absolute best care to the Artemis II crew.”
Aldridge, from Cushing, Okla., will be the first member to make contact with the crew upon their return to Earth. Assigned to Explosive Ordnance Disposal Expeditionary Support Unit 1, he will open the capsule, enter the space, and begin medical assessments.
“Coming from three generations of military service in my family, I’m honored to serve as the senior dive independent duty corpsman for this mission,” Aldridge said. “This effort is the culmination of both our training to bring world class care to the Artemis II crew and countless dedicated years of Navy Diving and Navy medicine.”
Dive independent duty corpsmen like Aldridge, Link, and Kapala are specifically trained in dive medicine.
Link, assigned to Explosive Ordnance Disposal Mobile Unit (EODMU) 1, is the third member of the team and hails from Chelsea, Mass. He has 18 years of experience in dive medicine, but he shares that this mission is already a highlight of his career.
“I have been exposed to the Navy since I was a young teenager, and I’m proud to represent both my family and hometown,” Link said. “Contributing our efforts to NASA and the Artemis II mission is something we take great pride in as part of that legacy.”
The fourth member, Kapala, assigned to EODMU-11, hails from Alpena, Mich., and has been practicing dive medicine since 2018. He notes the significance of the historical mission as a unified effort, both for himself and everyone involved.
“I grew up reading sci-fi novels and watching space movies, never thinking that I would play a part in a recovery mission like this,” Kapala said. “It is surreal to play a part in safely recovering the astronauts from the capsule to get them home safe to their families, an effort that really makes you realize this team is bigger than just the four of us.”
With immense levels of experience and specialized training under their belts, this team of expeditionary medical providers is prepared to give the Artemis II crew a warm welcome back to Earth following their 10-day lunar mission.
“Our fellow divers, the Sailors on the ship, the helicopter squadron, our partners at NASA, and everyone supporting this mission are ready to bring the Artemis II crew home,” Wang said. “This team is undoubtedly making history.”
U.S. Navy Divers assigned to EODGRU-1 are underway on USS John P. Murtha in the U.S. 3rd Fleet area of operations supporting NASA’s Artemis II mission, retrieving the crew and spacecraft following their return to Earth and splashdown in the Pacific Ocean. NASA’s Artemis II mission sent four astronauts on a flight around the moon in the Orion space capsule, marking the first time humans journeyed to deep space in over 50 years.
Story by Lt Erin Wiley
Explosive Ordnance Disposal Group One