LAS VEGAS, NV — Jan 20-23, 2026, Phokus Research Group will present a selection of integrated medical systems at SHOT Show 2026, exhibiting in the ADS Inc. Booth #32103 / Law Enforcement Hall, where the company will showcase scalable solutions spanning discreet everyday carry, individual trauma response, advanced surgical support, and realistic medical simulation products. In addition to the featured products, Phokus will debut more than 20 additional new products across its medical, training, and support product lines during the show.
The lineup reflects Phokus’ role as a medical systems integrator—bringing together in-house design, mission-driven configuration, and strategic manufacturing partnerships to deliver medical solutions built for real-world operational environments.
Discreet / EDC Medical Systems
Vertx Gamut 18L Sling – Phokus Trauma Configuration

Designed for environments where overt medical or tactical equipment is inappropriate, the Phokus Trauma Configuration of the Covert Aid Sling (CAS) provides discreet, organized medical readiness in a low-profile everyday-carry platform. Built on the Vertx Gamut 18L Sling chassis, the system enables rapid access to critical items for trauma management while maintaining a non-descript appearance suitable for civilian, corporate, and executive protection environments.

The configuration prioritizes mobility, intuitive layout, and immediate visual clarity during critical incidents. Offered in scalable trauma loadouts, it allows users to align medical capability with operational risk without changing platforms or compromising discretion—supporting prepared civilians and protection professionals operating in dynamic, public-facing settings.
Individual First Aid Kits (IFAKs) – Modular & Combat-Proven
ORK IFAK (Operator Response Kit)

The ORK IFAK (Operator Response Kit) is a Phokus-configured individual first aid kit built around the High Speed Gear Med Pak pouch, a battle-proven platform recognized for durability, modularity, and rapid access. By leveraging the Med Pak’s established design, the ORK provides a flexible foundation for organized medical carriage across a wide range of operational roles.
Phokus configures the ORK in accordance with Tactical Combat Casualty Care (TCCC) guidelines to support mission-driven medical readiness, emphasizing consistent access under stress and a customizable internal organization. Features include a quick-access pull tab, MOLLE compatibility, and a rigid internal organizer panel that is configurable with shock cord and elastic retention, allowing users to tailor the medical carriage while maintaining speed and reliability during self-aid or buddy-aid.
Manufactured by High Speed Gear in the United States and Berry-compliant, the ORK IFAK pairs a trusted pouch platform with Phokus’ configuration expertise to deliver a dependable, operationally relevant medical solution.
Medic & Role 1 Level Systems
Phokus Surgical Ruck (Patent Pending)

The Multi-Mission Medical-Surgical Ruck (M3R-SURG) is a collaboration between Phokus and High Speed Gear, engineered for damage control resuscitation (DCR) and damage control surgery (DCS). Developed to address the demands of advanced care for teams operating in austere, mobile, or forward environments, the system features an external aluminum frame to enhance load carriage. Unlike other solutions on the market, the Surgical Ruck was also designed to store personal sustainment gear (food, water, warming layers), critical for extended operations.

The Surgical Ruck is scalable, agile, and rapidly deployable. It features modular, clear pouches mounted on rigid back panels, enabling teams to configure layouts according to mission requirements. Intended for austere surgical sites, the Surgical Ruck enables the rapid setup of a DCR/DCS capability through internal hanging bags and instrument panels, supporting an efficient transition from movement to advanced care delivery.
Training & Simulation
Wound Torso – In-House Research & Development

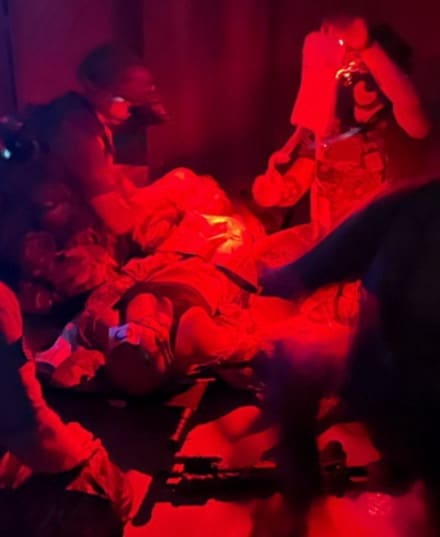
Phokus will also showcase its patent-pending Wound Torso task trainer, developed through in-house research and design to support realistic training in hemorrhage control and trauma management. The upper-body task trainer is anatomically configured to simulate complex, high-threat injuries commonly encountered in both military and civilian critical incidents, with emphasis on junctional and non-compressible hemorrhage.
Designed to reinforce TCCC and NREMT principles, the Wound Torso enables responders to train on anatomically relevant wound patterns, treatment priorities, and decision-making under stress. The modular wound cover options allow instructors to tailor scenarios to specific training objectives.
SHOT Show 2026
Phokus Research Group will be exhibiting at SHOT Show 2026 in ADS Inc. Booth #32103 / Law Enforcement Hall, where attendees can view these products in person and engage with the Phokus team on application, configuration, and system integration.